The response of ultrasound and clinical features of gout in patients managed with a treat-to-target serum urate protocol
David Bursill (1,2), Anita Lee1,2, Kim Griggs (1,2), Phoebe Hunter (1,2), Emma Hocking (1,2), Leah McWilliams (1), Vidya Limaye (1,2), Susanna Proudman (1,2)
Affiliation(s):
1. Rheumatology Unit, Royal Adelaide Hospital, Adelaide, South Australia, Australia.
2. University of Adelaide, Adelaide, South Australia, Australia
Background: Treat-to-target (TTT) urate lowering therapy (ULT) is an effective approach to the management of gout and ultrasound (US) is a sensitive tool to detect monosodium urate (MSU) crystal deposition. We investigated the progression and relationship between clinical and US-detected features of gout in patients undergoing TTT ULT in a protocolised outpatient clinic.
Methods: Patients over 18 years with gout (defined by 2015 ACR-EULAR classification criteria) was managed with a standardised TTT ULT protocol at the Royal Adelaide Hospital. Patients were assessed at baseline, 6 and 12 months for clinical features of gout (flares, subcutaneous tophi, tender and swollen joints and serum urate [SU]) and US features of gout (double contour sign [DCS], tophi, hyperechoic foci [HEF], erosions, effusion, synovitis and power Doppler positivity). US sites examined included the knees, peroneus tendons and all metatarsophalangeal (MTP) joints. Subcutaneous tophus volume (STV) was calculated (STV = [width2 × length]/2) based on measurements taken with digital Vernier callipers. US tophus burden was calculated by summing the maximal diameter of the two largest tophi detected at each site (three largest at the knees). US evidence of effusion and synovitis were graded 0 to 3; only moderate) and severe grades were used for subsequent analyses. Statistical analyses were performed using IBM SPSS Statistics (version 29.0.1.0). Data were transformed using ARTool for parametric analysis. Variable trends were evaluated using Friedman's ANOVA and Wilcoxon Signed Rank tests with Bonferroni-adjusted post hoc analysis, alongside a repeated measures ANOVA.
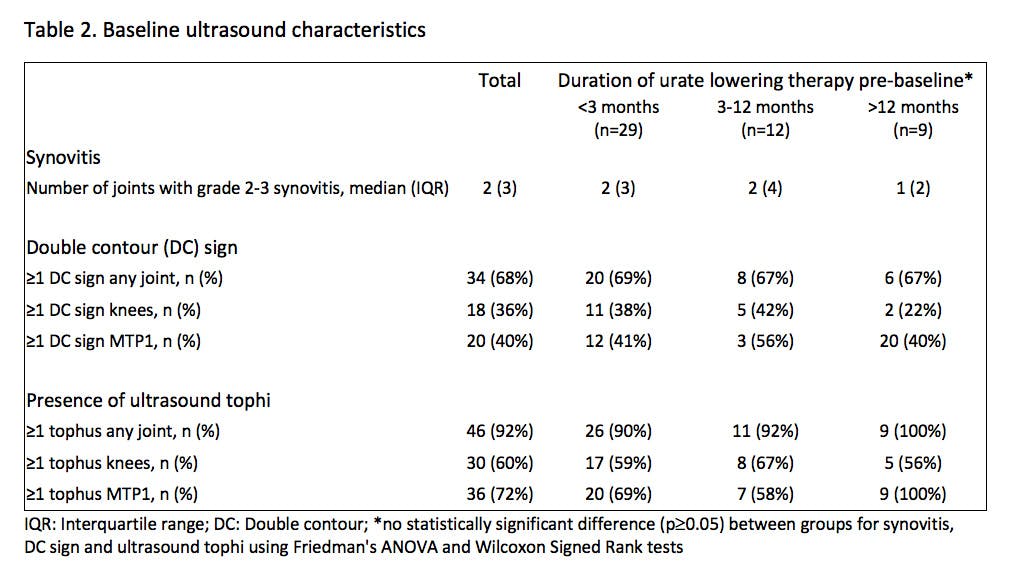
Results: 50 patients (82% male, mean age 62.9 ± 14.0 years, median disease duration 9.4 [IQR 24.2] years) were included (Table 1 and 2). At baseline, there was no significant effect of ULT duration on STV, DCS or US tophus burden. SU significantly decreased from baseline to month 6 (median 0.43 [IQR 0.18] to 0.30 [IQR 0.07] mmol/L, p<0.001), with 43.8% participants at 6 months and 60.9% at 12 months achieving the target of <0.30 mmol/L (p<0.001). Flare frequency was also significantly reduced by 6 months (median 0.42 [IQR 0.7] to 0.00 [IQR 0.05] flares/month, p<0.001). STV significantly decreased from baseline to month 6 (median 14.75 [IQR 42] to 0.96 [IQR 22] cm3, p=0.01, Figure 1). Correspondingly, US features of MSU crystal deposition also significantly reduced from baseline to month 6, including DCS (median 1 [IQR 2] to 0 [IQR 1] sites/patient, p=0.003) and US tophus burden (median 11.85 [IQR 22.1] to 8.95 [IQR 18.2] mm, p<0.001, Figure 1). US features of inflammatory arthritis did not decrease at 12 months, with no significant change in effusion (mean 7.4 [SD 2.1] to 7.4 [SD 2.6]), synovitis (median 2 [IQR 3] to 1 [IQR 1]) or power Doppler (median 0 [IQR 2] to 0 [IQR 1]) positive sites per patient (all p>0.05). Ultrasound was able to detect evidence of MSU deposition (≥1 tophus and/or DCS) in 27/28 (96%) patients with no clinically discernible subcutaneous tophi; limited ultrasound examination of the knees and first MTP joints detected MSU deposition in 24 (86%) of this subgroup with clinically non-tophaceous disease.
Conclusion: TTT ULT can lead to early improvements in the clinical features of gout, including flares and subcutaneous tophi, which corresponds to the temporal reduction of MSU crystal deposits detected on US. US evidence of inflammation persisted at 12 months which has implications for the disease course of this chronic inflammatory arthritis and may influence treatment decisions. US provides a valuable, non-invasive assessment of the subclinical manifestations of gout and their response to ULT.